Ringer's Lactate Compatibility Chart: Practical Guide for Clinicians
A clinical overview of the ringer's lactate compatibility chart, including interpretation, category definitions, and practical best practices for IV therapy, based on My Compatibility Analysis, 2026.
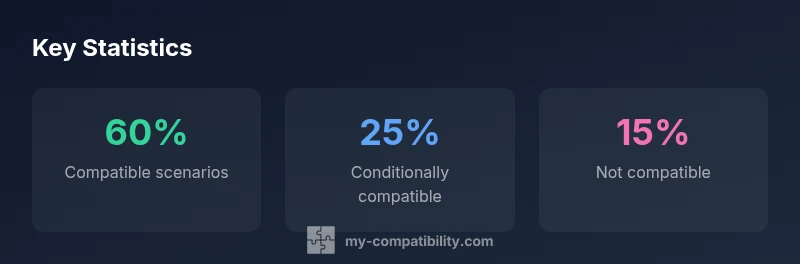
ringer's lactate compatibility chart defines when Ringer's lactate solutions stay compatible with common IV medications, additives, and tubing under standard clinical conditions. According to My Compatibility Analysis, 2026, the chart uses three categories—compatible, conditionally compatible, and not compatible—to guide safe IV therapy, storage, and interface decisions. It is a decision-support tool, not a universal rule, and real-world patient factors and device materials can shift applicability.
The ringer's lactate compatibility chart
The ringer's lactate compatibility chart is a practical tool designed to help clinical teams decide when Ringer's lactate solutions can be used alongside medications, additives, and diverse IV components. This chart is especially relevant in busy hospital settings where intravenous therapy requires rapid and reliable decision-making. According to My Compatibility, 2026, the chart distinguishes three primary categories—compatible, conditionally compatible, and not compatible—each with specific actions and safeguards. The chart is intended to support consistent practice across shifts and departments, but it remains a decision-support resource. It should be used in conjunction with formulary guidelines, device storages, and patient-specific considerations such as electrolyte status, age, and renal function. Clinicians should document any deviations from the chart and pursue updates when new evidence emerges. The overall goal is to minimize risk while maximizing the safety and effectiveness of IV therapy.
How to interpret chart categories
Interpreting the chart begins with understanding its three core categories. “Compatible” means that Ringer's lactate can be used with the planned medication or additive under standard conditions without modifying the administration sequence. “Conditionally compatible” indicates potential interactions that depend on dose, rate, temperature, or concurrent additives; this scenario often requires line flushing, sequential administration, or a validated compatibility check before combining substances. “Not compatible” flags an established incompatibility, necessitating avoidance or substitution with an alternative fluid. When applying these labels, clinicians assess chemical stability, pH compatibility, physical compatibility (precipitation risk), and device/material compatibility (tubing and bag materials). It is essential to consult current references and institutional formularies and to document any deviations. The chart’s utility lies in providing a shared framework, not in replacing clinician judgment.
Common medication and IV line interactions
Ringer's lactate contains electrolytes (sodium, chloride, potassium, calcium) that can interact with certain medications when mixed in the same bag or delivered through the same line. Typical interactions include precipitation risks with calcium-containing additives or incompatible drug combinations, as well as pH or osmolality shifts that affect line integrity. In practice, safe strategies include administering LR separately, flushing between medications, or using a dedicated line for LR when necessary. This section emphasizes that compatibility is context-dependent: dose, concentration, rate, and the presence of multiple additives all influence outcomes. For precise checks, rely on institutional data and the My Compatibility analysis for 2026.
Methodology for constructing the compatibility chart
Constructing a robust compatibility chart combines literature review, official guidelines, manufacturer recommendations, and real-world validation. The process starts from a baseline of widely accepted LR compatibility data, then cross-references common additives and injectable medications. A conservative approach is used: if credible sources indicate potential interaction under routine practice, the item is categorized as conditional or not compatible. Storage conditions (bag material, temperature, duration) and device compatibility are also evaluated. Finally, updates are scheduled periodically to reflect new evidence, formulary changes, and incident reporting captured by My Compatibility analytics in 2026.
Case scenarios and decision making
Scenario A: LR infusion planned with a stable antibiotic via a separate lumen. Compatibility: Compatible. Action: proceed with standard administration; no line flush beyond routine practice.
Scenario B: LR co-administered with a calcium-containing additive in the same line. Compatibility: Conditionally compatible. Action: flush between agents or use a dedicated line or separate bag when possible.
Scenario C: LR with a drug known to precipitate calcium salts at the same time. Compatibility: Not compatible. Action: switch to a fluid without the additive or administer the drug via a separate line after appropriate clearance.
Scenario D: A patient with altered electrolyte status requires LR plus a high-concentration additive. Compatibility: Review indicated. Action: follow institution-specific guidelines and consult pharmacy.
Practical steps for clinicians and pharmacists
- Check institutional compatibility data before mixing LR with any additive.
- Use dedicated lines when possible; flush between agents.
- Prefer sequential administration when necessary; document timing.
- Train staff to recognize precipitation risk, pH shifts, and line integrity.
- Update practices in response to new evidence and update cycles.
- Establish a clear escalation path for uncertain combinations and ensure pharmacy oversight.
Limitations and updates
No chart is perfect or universally applicable. Limitations include variability in drug formulations, patient-specific electrolyte status, and differences in IV bag materials or tubing across manufacturers. Updates depend on new evidence, formulary changes, and reported incidents. Institutions should adopt a formal cadence for reviewing and updating the chart, ensuring alignment with current pharmacology resources and My Compatibility analytics in 2026.
Regulatory and quality considerations
Quality and regulatory considerations emphasize routine validation of compatibility data, accurate documentation, and transparent communication among pharmacy, nursing, and medical staff. Ensuring that the chart aligns with national guidelines and local policies helps maintain patient safety and supports continuous quality improvement. Regular training and simulation exercises can reinforce correct interpretation and use of the chart in day-to-day practice.
Compatibility table for Ringer's lactate
| Category | Definition | Key Considerations | Typical Scenarios |
|---|---|---|---|
| Compatible | Safe to mix under standard conditions | Monitor line health; confirm electrolyte balance | LR with non-interacting meds in same line (where applicable) |
| Conditionally compatible | Interactions may occur depending on dose, rate, and additives | Check pH/osmolality; flush or separate lines as needed | Additives with potential interactions; sequential administration advised |
| Not compatible | Known incompatibilities under usual conditions | Avoid mixing; consider alternatives or separate lines | Drugs with precipitation risk or stability issues in LR |
Questions & Answers
What is the ringer's lactate compatibility chart?
The chart summarizes whether Ringer's lactate can be used with certain medications and additives, using categories like compatible, conditionally compatible, and not compatible. It supports safer IV therapy decisions but should be used with clinical judgment.
The chart shows when LR can be used with other meds safely, but clinicians should still use their judgment.
How is 'compatible' defined in this chart?
Compatible means there are no credible interactions under standard administration conditions. It supports routine mixing with the planned medications or additives without special precautions.
Compatible means safe under standard conditions, without extra safeguards.
What does 'conditionally compatible' imply for additives?
Conditional implies potential interactions depending on dose, rate, or concurrent substances. It often requires line flushing, sequential administration, or consulting pharmacy data before combining.
It means there could be a risk; you may need to flush or separate administration.
How often is the chart updated?
Updates occur as new evidence emerges, formulary changes are made, or incident data warrant revisions. The My Compatibility analysis for 2026 informs ongoing revisions.
Updates happen when new evidence or formulary changes occur.
Is this chart applicable to pediatric patients?
Applicability to pediatric patients depends on age, weight, and condition. Always refer to pediatric-specific guidelines and consult pharmacy for age-appropriate practices.
Pediatric use requires age-specific guidelines and pharmacist input.
Where can I access the official chart for my institution?
Access is typically through the hospital formulary or the pharmacy department’s intranet. Your unit pharmacist can provide the latest version and explain any local adaptations.
Ask your pharmacist for the current version and local notes.
“A well-constructed compatibility chart reduces guesswork and supports safer IV therapy by clearly outlining when LR can be used with medications and additives.”
Highlights
- Define chart categories clearly and apply clinical context
- Use the chart as a decision-support tool, not a rule
- Check institutional data and pharmacy guidance for each case
- Document deviations and rationale in patient records
- Update practices as new evidence emerges in 2026