IV Medication Compatibility Checker: Safety & Interactions
A comprehensive guide to IV medication compatibility checkers, explaining how they assess interactions, dosing, and device compatibility to improve infusion safety in hospital and home settings.
An IV medication compatibility checker is a decision-support tool that analyzes active ingredients, excipients, diluents, buffers, and infusion protocols to flag potential incompatibilities, interactions, and precipitation risks. By evaluating medication compatibility across bag, line, and device configurations, it helps clinicians and caregivers choose safe combinations, adjust dosing, or select alternative administration strategies.
What the IV Medication Compatibility Checker Does
The IV medication compatibility checker is a decision-support tool designed to help clinicians and caregivers reduce infusion risks. At its core, it analyzes active ingredients, excipients, diluents, buffers, and administration routes to identify potential incompatibilities. It flags situations that could lead to precipitation, chemical reactions, or unsafe interactions when meds share a common line, bag, or device. The tool can also note when additive regimens require changes in concentration, pH, or time sequencing. By providing a structured risk assessment, it supports safer IV therapy across hospital wards, outpatient clinics, and home infusion programs. In practice, users enter the meds and diluents planned for a given infusion and review a clearly labeled set of alerts with recommended actions. The goal is to prevent adverse events before they occur, while preserving therapeutic efficacy. This checker is most effective when deployed as part of standard ordering and compounding workflows, with updates synchronized to the latest formularies and evidence databases.
How Checks Are Performed: Data, Rules, and Sources
Checks rely on a blend of data sources and logic. Authoritative drug databases, institutional formularies, and vendor compatibility citations feed the risk models, while clinical guidelines shape recommended actions. The checker uses rule-based logic to classify alerts by severity and to suggest concrete steps—such as adjusting concentration, changing diluents, or sequencing doses. It also incorporates patient-context inputs (age, renal function, electrolyte status) when available, to tailor alerts to the individual. The underlying algorithms balance sensitivity and specificity: too many false alarms can erode trust, while missed risks threaten safety. To maintain accuracy, the system logs every decision and periodically audits alerts against adverse event reports. All outputs are designed to be explainable, with concise rationale for each warning and links to supporting references. It is important to note that these tools augment, not replace, clinical judgment.
Dosing, Diluent, and Infusion Considerations
IV compatibility is not only about the active drug but also how it is prepared and delivered. The checker accounts for diluents (saline, dextrose), concentrations, and infusion rates because these factors influence stability. It flags when a diluent is incompatible with a medication’s formulation or when two drugs might interact within the same bag or line. It also highlights container-related issues, such as compatibility with IV bags, administration sets, and additives that affect pH or ionic strength. In pediatrics and neonatology, small-volume infusions and compounded mixtures introduce additional complexity; the tool provides tiered guidance based on patient age and weight. Practically, a nurse or pharmacist can review the alerts, adjust the preparation plan, and confirm that the final infusion remains within stability and compatibility guidelines before administration.
Cross-Compatibility Across Bags, Lines, and Sets
The IV medication compatibility checker evaluates not just the drug–drug pair but the entire infusion chain. It assesses bag-to-line interfaces, connector types, and the potential for interactions when two meds share a common lumen. It also considers carrier solutions (e.g., saline vs dextrose) and the possibility of precipitation at different temperatures or storage times. For devices with multiple Y-sites or split infusions, the checker maps the flow path to determine where risk might emerge. The result is a practical set of actions: remove a drug from the same line, switch to a separate site, substitute with a compatible alternative, or schedule sequential dosing with adequate flushing. In routine practice, this holistic view helps prevent incompatibilities that could otherwise go unnoticed during busy rounds.
Practical Workflow: From Prescription to Infusion
In real-world settings, the tool is most valuable when embedded in the clinical workflow. Prescribers or pharmacists select the IV medications and diluents, then the compatibility checker runs automatically or on-demand. The results appear as a prioritized list of alerts, with high-severity issues at the top and practical remediation steps beneath. Nursing staff use these recommendations at bedside, verifying dosages, preparing mixtures under aseptic conditions, and confirming line configuration before starting the infusion. The system keeps an audit trail, timestamps, and links to references used for each warning. When conflicts arise, teams can rapidly sequence therapies, adjust infusion times, or choose alternative agents with similar therapeutic profiles. Regular training helps keep staff confident in interpreting alerts and applying the recommended actions.
Common Pitfalls and Risk Mitigation
No system is perfect, and several pitfalls can undermine the usefulness of a checker if not managed. Outdated formularies and unsupported drug entries lead to missed risks. Incomplete patient context (e.g., renal function, critical illness) can yield less relevant alerts. Alert fatigue remains a major challenge; clinics should tune severity levels and maintain a short, actionable set of warnings. Data quality matters: incorrect lot numbers or mislabeled diluents can propagate false alerts. Finally, human factors—hand-off communication, workload, and interruptions—can erode safety if a checker is treated as a substitute for professional judgment. The best mitigation combines regular data updates, pharmacist oversight, and routine drills that simulate real infusion scenarios.
Integrating with Pharmacy and EHR Systems
Seamless integration with pharmacy information systems, electronic health records (EHR), and infusion therapy modules is essential for widespread adoption. Interoperability standards, such as HL7 or FHIR-based interfaces, enable real-time data exchange of medications, dosages, and patient context. A well-integrated checker surfaces alerts within the clinician’s existing workflow—order entry, pharmacy compounding notes, and bedside documentation—reducing the need to switch between tools. Data provenance and audit trails support accountability, while configurable roles ensure the right team members see the appropriate alerts. Security and privacy controls protect patient data, and regular vendor updates keep the knowledge base current. Training and change management are critical: staff should understand how alerts are generated and how to apply the suggested remediation without delaying care.
Future Trends: AI, Real-World Data, and Regulation
As data resources grow, IV compatibility tooling is likely to incorporate machine learning to refine risk predictions and personalize recommendations. Real-world data from infusion centers can validate and expand the knowledge base beyond controlled studies. Regulatory oversight may require transparent model explanations and periodic revalidation of alert logic. The My Compatibility team expects ongoing improvements in user experience, with more intuitive dashboards, inline documentation, and smarter default settings that adapt to a clinic’s practices. Clinicians can anticipate tighter integration with pharmacy workflows and more proactive alerts that help prevent adverse events before they happen.
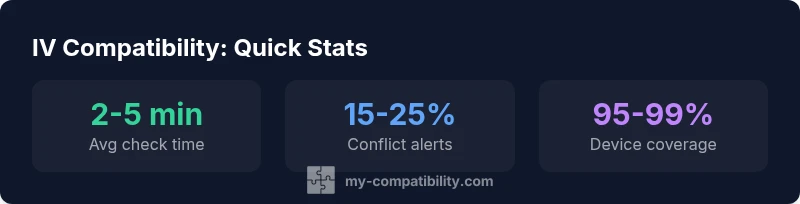
Key performance metrics for IV compatibility checking
| Metric | Range | Notes |
|---|---|---|
| Check time | 2-5 minutes | Average time to run a check |
| Conflict alerts | 15-25% | Estimated alerts per check |
| Device coverage | 95-99% | Infusion device compatibility spread |
| Clinician adoption | 40-60% | Current usage in pilot sites |
Questions & Answers
What is an IV medication compatibility checker?
An IV compatibility checker analyzes active ingredients, diluents, and infusion configurations to flag incompatibilities and precipitation risks before drugs are mixed or infused.
An IV compatibility checker analyzes meds and diluents to flag incompatibilities before infusion.
Can these tools replace a pharmacist’s review?
No. These tools augment pharmacist judgment by highlighting potential risks, but final decisions should involve a pharmacist, especially for complex regimens or high-risk patients.
They don’t replace pharmacists; they support safer decisions.
What data sources do they rely on?
They rely on authoritative drug databases, institutional formularies, provider notes, and current guidelines to inform alerts and remediation steps.
They use trusted drug databases and formularies to flag risks.
Are IV compatibility checkers suitable for home infusion settings?
Yes, when paired with clear guidance and access to a clinician, these tools can help caregivers manage injections and infusions safely at home.
They can help with home infusions, with proper support.
How should alerts be interpreted and acted upon?
Treat alerts as safety signals. Review severity, verify patient context, and apply recommended actions such as dose adjustments or alternative regimens before proceeding.
Take alerts seriously and follow the recommended steps.
Do these checkers cover all IV bags and devices?
Most checkers cover common bags, lines, and sets, but gaps may exist for rare devices or new formulations. Regular updates help close these gaps.
They cover common setups, but updates are essential for new devices.
“A robust IV compatibility checker reduces risk by surfacing interactions before they reach the patient.”
Highlights
- Verify IV compatibility before administration with checks that consider meds and diluents
- Expect 2-5 minute checks in typical workflows
- Alerts often reflect 15-25% of checks, highlighting high-severity risks
- Integrate checks into ordering and compounding workflows for efficiency
- Regular updates and clinician oversight sustain safety and trust